by Sharon Rondeau
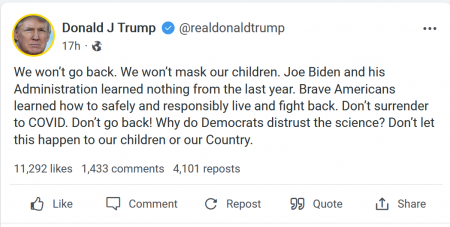
(Jul. 28, 2021) — Following an announcement by the Centers for Disease Control and Prevention (CDC) that it was issuing new “guidance” that both COVID-19-vaccinated and non-vaccinated individuals revert to wearing masks indoors in areas “of substantial or high transmission” as well as reports that Oval Office occupant Joe Biden is likely to issue an order for federal employees to be vaccinated or tested weekly as a condition of employment, 45th President of the United States Donald J. Trump responded by accusing “Democrats” of “distrusting the science” and urging Americans not to “surrender to COVID.”
“We won’t go back. We won’t mask our children,” Trump said in a statement released late Tuesday. “Joe Biden and his Administration learned nothing from the last year. Brave Americans learned how to safely and responsibly live and fight back. Don’t surrender to COVID. Don’t go back! Why do Democrats distrust the science? Don’t let this happen to our children or our Country.”
The CDC recently recommended all children over the age of 2 wear masks in inside spaces. Presently, children under the age of 12 are not eligible for any coronavirus vaccine. At the time of its declaration on schoolchildren and masking (July 9), the CDC emphasized mask mandates for those not “fully vaccinated”; however, the new guidance issued Tuesday does not differentiate between unvaccinated and vaccinated children.
Children are generally not considered susceptible to severe illness from the virus, and some physicians believe they can actually transmit antibodies to others if they have been infected, knowingly or unknowingly.
Trump, who survived the virus during the closing weeks of the presidential campaign prior to the availability of the vaccines, in March 2020 agreed to a 15-day shutdown recommended by his coronavirus task force to “slow the spread” and avoid overwhelming hospitals.
Trump was quick to express cautious optimism about the country reopening by mid-April that year, but the shutdown continued for months, varying from one part of the country to another. While majority-“red” states tended to reopen their economies sooner and abandon mask mandates as the unprecedented situation unfolded, most “blue” states continued mask mandates in public places and partial closures or restrictions.
Earlier this year, the CDC issued guidance stating that vaccinated individuals could forego masking indoors, leading many establishments to drop the requirement.
While it was unfolding and after leaving office, Trump touted his “Operation Warp Speed” which funded coronavirus vaccines n record time by Moderna, Pfizer and Johnson & Johnson. Trump has recommended that Americans take the vaccine but also acknowledged that they should be able to make an informed decision.
Emergency Use Authorization (EUA) for coronavirus vaccines emerging from Operation Warp Speed was granted late last year. In November, the FDA explained the designation:
An Emergency Use Authorization (EUA) is a mechanism to facilitate the availability and use of medical countermeasures, including vaccines, during public health emergencies, such as the current COVID-19 pandemic. Under an EUA, FDA may allow the use of unapproved medical products, or unapproved uses of approved medical products in an emergency to diagnose, treat, or prevent serious or life-threatening diseases or conditions when certain statutory criteria have been met, including that there are no adequate, approved, and available alternatives. Taking into consideration input from the FDA, manufacturers decide whether and when to submit an EUA request to FDA.
As reported by the CDC, COVID-19 vaccines are “a new type of vaccine to protect against infectious diseases. To trigger an immune response, many vaccines put a weakened or inactivated germ into our bodies. Not mRNA vaccines. Instead, they teach our cells how to make a protein—or even just a piece of a protein—that triggers an immune response inside our bodies. That immune response, which produces antibodies, is what protects us from getting infected if the real virus enters our bodies.”
Further CDC explanation of how mRNA vaccines work reads:
mRNA vaccines have been studied before for flu, Zika, rabies, and cytomegalovirus (CMV). As soon as the necessary information about the virus that causes COVID-19 was available, scientists began designing the mRNA instructions for cells to build the unique spike protein into an mRNA vaccine.
Future mRNA vaccine technology may allow for one vaccine to provide protection for multiple diseases, thus decreasing the number of shots needed for protection against common vaccine-preventable diseases.
Beyond vaccines, cancer research has used mRNA to trigger the immune system to target specific cancer cells.
The CDC does not say that no mRNA vaccines have yet been approved by the FDA.
The media and “Big Tech” have attempted to dispel any negative aspects of the vaccines, with YouTube, Facebook and Twitter censoring content questioning their efficacy and/or safety.
Trending topics on Twitter on Wednesday include “People should still get vaccinated even if they’ve already had COVID-19, health agencies and medical experts say.”
Dr. Robert Malone, who invented the mRNA technology but has cautioned about possible risks from widespread coronavirus vaccination, has been censored by YouTube and received his “first threat,” he wrote on Twitter Monday.
On July 26, Reuters reported:
Most people with allergic reactions to the first dose of an mRNA COVID-19 vaccine from either Pfizer/BioNTech or Moderna can safely receive the second dose, a new study shows. The rates of allergic reactions to these vaccines have been reported to be as high as 2%, with anaphylaxis, the most serious kind, occurring in up to 2.5 of every 10,000 vaccine recipients, the researchers said. They reviewed data on 189 adults with first-dose reactions to one of these vaccines, such as flushing, dizziness or lightheadedness, tingling, throat tightness, hives, and wheezing or shortness of breath. Most of these adults – 84% – received the second dose of the vaccine, with about a third taking an antihistamine beforehand. All of them tolerated the second dose, including those with first-dose anaphylactic reactions. Any potentially allergic symptoms that developed after the second dose were mild and easily controlled, the researchers reported on Monday in JAMA Internal Medicine https://bit.ly/3iSeJLI. “Complete two-dose vaccination has become even more important with the Delta variant and we suspect there are many more people who did not get their second shot because of allergic symptoms,” said coauthor Dr. Matthew Krantz from Vanderbilt University. “Our data suggest that most patients with immediate and potentially allergic reactions to mRNA COVID-19 vaccines tolerate a second dose,” his team concluded.
According to the Mayo Clinic, “anaphylaxis” “can be fatal” and is defined as:
a severe, potentially life-threatening allergic reaction. It can occur within seconds or minutes of exposure to something you’re allergic to, such as peanuts or bee stings.
Anaphylaxis causes your immune system to release a flood of chemicals that can cause you to go into shock — your blood pressure drops suddenly and your airways narrow, blocking breathing. Signs and symptoms include a rapid, weak pulse; a skin rash; and nausea and vomiting. Common triggers include certain foods, some medications, insect venom and latex.
Anaphylaxis requires an injection of epinephrine and a follow-up trip to an emergency room. If you don’t have epinephrine, you need to go to an emergency room immediately. If anaphylaxis isn’t treated right away, it can be fatal.
“Pfizer says 2021 COVID-19 vaccine sales to top $33.5 billion, sees need for boosters,” Reuters reported Wednesday, a claim the CDC refuted on July 8.
In 2014, Reuters CEO James C. Smith was appointed to Pfizer’s Board of Directors, a position he still holds, according to Pfizer’s website.
As of May 30, 2021, approximately half of all U.S. adults had received a vaccine, the CDC reported. “The findings in this report indicate that trust in COVID-19 vaccines, particularly in their safety and effectiveness, was an important factor in the decision to get vaccinated among adults aged 18–39 years, especially for those who were unsure about or probably planning on getting vaccinated,” the report additionally states.
On Tuesday the group America’s Frontline Doctors (AFLDS) gave an hours-long summit, the third of its kind in a year, focusing on “the Science” surrounding the declared COVID-19 pandemic. The organization is rooted in the concept of freedom of speech and medical choice, including whether or not to take one of the coronavirus vaccines approved under the FDA’s EUA.
One Oregon physician who spoke, Dr. Philip LaTulippe, lost his medical license, livelihood and home after state health authorities, following an investigation into his non-requiring of masks by his staff and continuing to see patients in person, suspended, then revoked his medical license.
For treatments falling under an EUA, the CDC provides “Fact Sheets” to inform the public of the treatment’s alleged “risks and benefits.” The Pfizer Fact Sheet begins:
You are being offered the Pfizer–BioNTech COVID–19 Vaccine to prevent Coronavirus
Disease 2019 (COVID–19) caused by SARS–CoV–2. This Fact Sheet contains
information to help you understand the risks and benefits of the Pfizer–BioNTech
COVID–19 Vaccine, which you may receive because there is currently a pandemic of COVID-19.
However, at the bottom of the page 1 is the statement, “There is no FDA–approved vaccine to prevent COVID–19.”
AFLDS believes that the COVID-19 vaccines are “unsafe to widely distribute” because of their “experimental” nature and that the CDC should withdraw its emergency use authorization (EUA) for individuals age 30 and under due to severe side effects reported to the VAERS system hosted by the Department of Health and Human Services (HHS).
VAERS was established in 1990 and, according to data analyzed and presented at Tuesday’s AFLDS presser, has received more reports of adverse events, including deaths, in the approximately six months of COVID-19 vaccine administration than from all other vaccines in the U.S. combined since its inception.
Of the VAERS system, the CDC states:
Despite its limitations, VAERS is useful in that it generates signals that trigger further investigations. VAERS can detect unusual increases in previously reported events. As noted earlier, the sentinel role of VAERS is particularly significant for newly licensed vaccines, as evidenced in 2009 by the VAERS publication of the first summary of postlicensure H1N1 pandemic influenza safety data.[23] Although manufacturers are now routinely asked to conduct or sponsor postlicensure studies designed to collect additional safety data for large numbers of vaccine recipients, the need for a national postlicensure surveillance system remains. Like prelicensure studies, postlicensure studies may not be large enough to detect novel very rare AEs or may take several years to accumulate enough data to assess a rare occurrence.
At the end of June, Sen. Ron Johnson (R-WI) held a press conference in Milwaukee at which a half-dozen individuals reported serious injuries and disabilities attributed to the vaccines, including a 13-year-old girl who is now unable to consume food normally and is wheelchair-bound.
Because of the experimental nature of the vaccines, those claiming injuries are generally unable to file civil lawsuits for damages against the manufacturers.
AFLDS opposes medical “censorship,” including what it said occurred around the well-established drug hydroxychloroquine and other remedies for early treatment of the virus.


Dr. Peter McCullough, Vice Chief of Internal Medicine at Baylor University Medical Center, has also raised concerns over the CDC’s universal emphasis on vaccines as treatment for the coronavirus, citing hydroxychloroquine and other medications and protocols he maintains are effective against the disease, particularly if administered early.
In recent weeks and days, ostensibly due to the emergence of the “Delta” coronavirus variant, a number of businesses and public agencies, including Capitol Hill, have announced vaccine or mask “mandates,” with some in the mainstream media applauding and even encouraging such actions.
Citing a single source, CNN reported Tuesday that Biden “will announce on Thursday a requirement that all federal employees and contractors be vaccinated against Covid-19, or be required to submit to regular testing and mitigation requirements,“
According to NaturalHealth365 on July 23, there have been “nearly 11,000 deaths and 50,000 serious injuries in just 7 months of mass experimental injections.”
As AFLDS reported, a federal lawsuit it filed July 19 in the Northern District of Alabama against HHS claims, via a CDC whistleblower, seeks to “stop Emergency Use Authorization (EUA) of the experimental COVID-19 injections for three groups of Americans.”
“A knowledgeable AFLDS source inside the Centers for Disease Control (CDC) stated that deaths occurring within 72 hours of COVID-19 shot injection are significantly underreported in the publicly-available VAERS database maintained by the Federal Drug Administration (FDA),” the lawsuit states. “’This is shocking,’ the source said, ‘and informed consent, which is required under the law, is impossible when safety data is so misleading.’ As of July 9th, reported deaths in VAERS totaled 10,991. Of those, 4,593 were within 72 hours of vaccination. The CDC source has provided a sworn statement that the actual number of deaths are conservatively five times higher, meaning that as of publication, we are approaching at least 55,000 vaccine-related deaths.”


Thank God for COVID-19, the new gift that keeps on giving, that also eliminated seasonal flu and that annoying common cold.